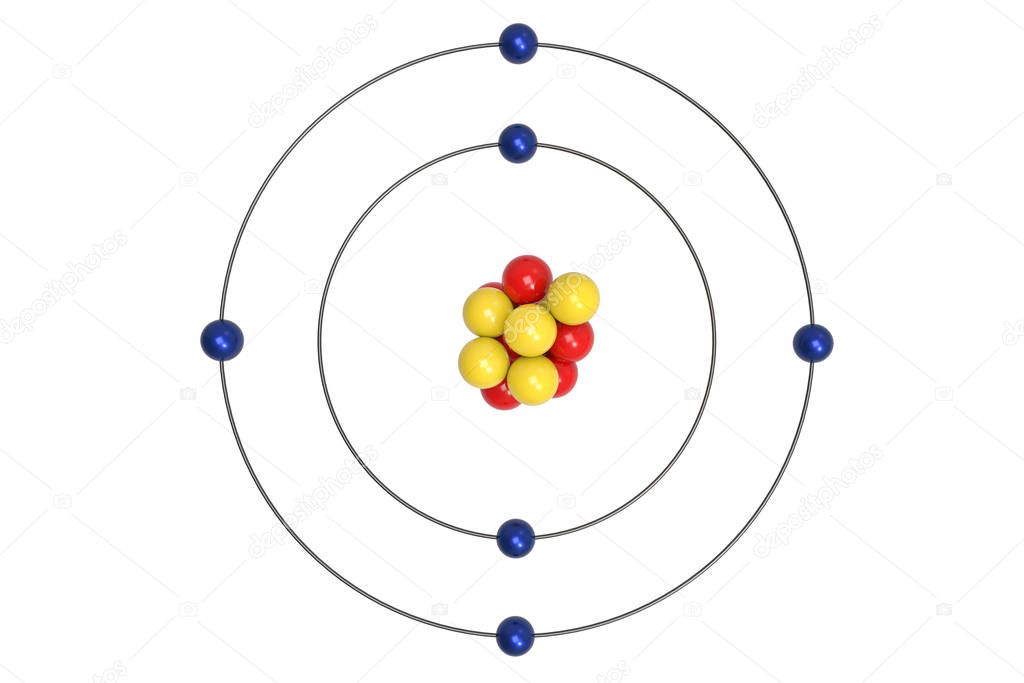
Bohr’s atomic model explains the stability of an atom on the basis that electrons energy remain constant in an orbit or shell.The difference in energy between two energy state is hv i.e. An elctrons jumps from higher energy level to lower energy level when it absorbs energy, while looses energy when the transition occurs from higher energy level to lower energy level.This postulates suggest that electron’s angular momentum is quantized Only those orbits are permitted whose angular momentum is the integral multiple of h/2π i.e.The energy of the electrons in a particular energy level remains constant, and hence accounts for the stability of an atom.The orbits are reprsented as 1, 2, 3, ….etc., and the shells are represented by K, L, M, N,……….etc. In an atom, electrons revolve around positively charged nucleus in a fixed cicular path called orbits or shells.The main postulates of Bohr’s model of the Hydrogen atom are: Bohr’s atomic model suggests that electrons surround the nucleus moving in orbit like planets around the sun. Postulates of Bohr’s model of atomīohr’s atomic model explains the structure of atoms, basically of the Hydrogen atom, and is therefore also called Bohr’s model of Hydrogen atoms. According to Bohr’s atomic model, “ a positively charged nucleus is surrounded by electrons moving in fixed orbits.” Later on, He was also awarded by Nobel prize in physics for his contribution to the structure of atoms. This atomic model is the modification of Rutherford’s atomic model (the nucleus is positively charged and is surrounded by electrons (negatively charged particles). In 1913, Danish physicist Neil Bohr proposed the Bohr atomic model based on Planck’s quantum theory of radiation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
